
News
In 2020, it was rated as a national specialized and new "little giant" enterprise by the Ministry of industry and information technology
2023-05-06 423
I. Background
In today’s world, oil shortages and white pollution are becoming increasingly severe, and people’s environmental awareness is gradually increasing. Bio-based degradable plastic polylactic acid (PLA) is polymerized by fermentation of raw materials such as corn to produce lactic acid. Due to its recyclability of raw materials, repeated thermal processability, and excellent mechanical and gas barrier properties compared to other bio-based plastics, it has become recognized as the most promising alternative to petroleum-based plastics.
As PLA synthesis technology continues to advance, its production costs continue to decrease. Due to its mechanical strength is similar to the existing plastic properties, it has a melting point of up to 140~180 °C, and because of its excellent transparency, it has gradually entered the fields of textile, tableware and product packaging from the biomedical field for application. However, due to the low nucleation density and crystallization rate of PLA, the general molded products are amorphous, which greatly reduces its heat resistance, seriously affects its mechanical, thermal processing and heat resistance properties, and greatly limits its application in electronics, electrical appliances and engineering plastics. Therefore, improving crystallization performance has become the key to broadening the application field of PLA.
Nucleation modification is an effective method to increase the crystallinity of PLA and accelerate the crystallization rate to improve the crystallization performance. Although the traditional petroleum-based plastics nucleating agents such as talc, zinc phenylphosphate and organic amide nucleating agents can also improve the crystallization ability of PLA to a certain extent, however, due to the nucleating agent itself is a non-organic biomass system, the addition and use of such nucleating agents destroy the biodegradable and recyclable characteristics of PLA to a certain extent.
Therefore, today, with the increasing demand for the application of green and environmentally friendly materials, it is urgent to choose a green biodegradable addition system to prepare “Double Green” biodegradable materials.
Ⅱ. Crystallization of PLA
Due to the presence of chiral carbon atoms, lactic acid has two conformations, D and L type. and two lactic acid molecules can be condensed to obtain three conformations of lactide, D, L, and DL type.
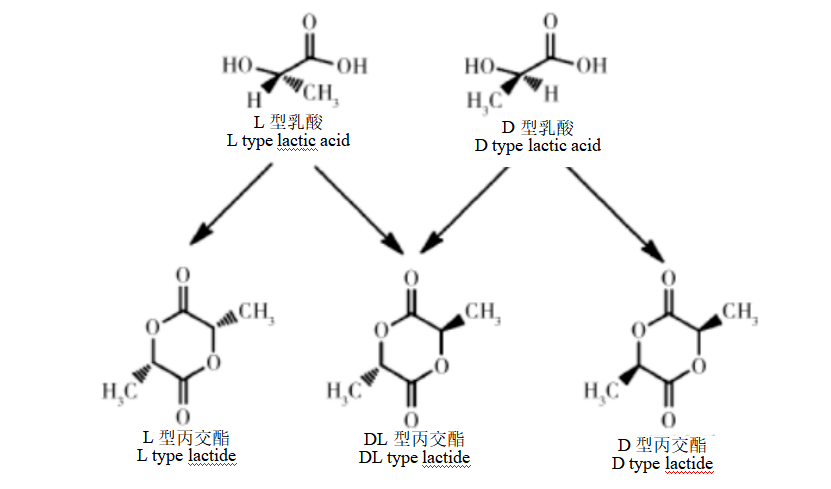
Figure 1: Structure of lactide and lactic acid
PLA is mainly obtained by ring-opening polymerization of lactide, so PLA has three optical isomers: poly-L-lactic acid (PLLA), poly-D-lactic acid (PDLA) and poly-D,L-lactic acid (PDLLA).
Due to different crystallization conditions, PLA exists in three crystal forms: α, β and γ. α crystal form is the most common and thermodynamically stable crystal form. Β crystal form can be obtained at higher tensile ratio (10~20) and tensile temperature (180~200°C). It is found that β crystal form of PLA has excellent impact resistance and heat resistance, and compared with α crystal, the β crystal is unstable and the melting point is about 10°C lower. γ crystal form is obtained by PLLA at around 140°C by epitaxial growth of hexamethylbenzene.
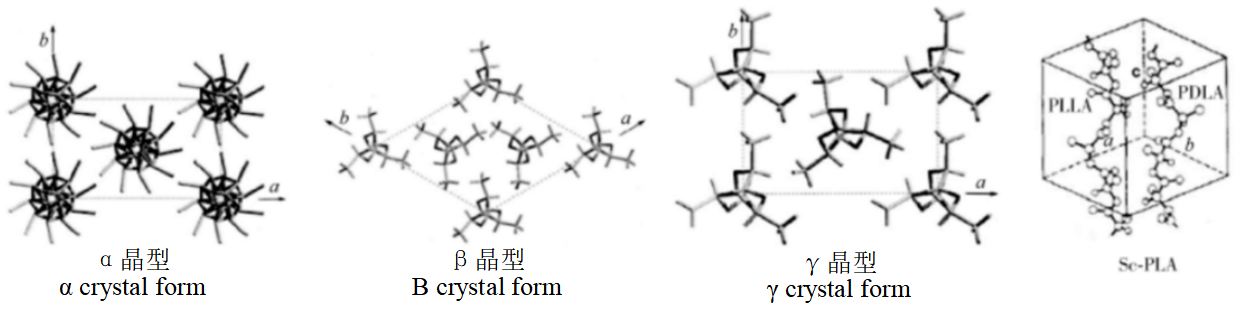
Figure 2: PLA crystal form structure model
Both PLLA and PDLA are in α crystal form, but stereoscopic composite crystal Sc-PLA can be obtained by blending PLLA and PDLA in a molten state of solution by equimolar ratio, which was first proposed by Ikada in 1987.
An important feature of the SC crystal form of PLA is that its melting point is approximately 50°C higher than that of the respective homopolymer. In addition, studies have shown that Sc-PLA can serve as its own nucleating agent to promote PLA crystallization.
Ⅲ. Nucleation Mechanism
At present, there are two main theories about the nucleation mechanism of PLA: Binsbergn’s heterogeneous nucleation mechanism and Wittmann’s epiphytic crystallization theory. Binsbergn believes that nucleating agents are used as heterogeneous crystal nuclei, and the surface of the non-polar part will be dented, and the molecular chains of the polymer will be neatly arranged in it, promoting the crystallization process of the polymer. Epiphytic crystallization refers to unidirectional or multidirectional crystallization of a crystalline substance on its surface based on another crystal.
Ⅳ. Organic biomass nucleating agent for PLA use
1. Polysaccharides
Starch: Due to the large amount of hydroxyl groups, starch has poor compatibility with PLA in practical applications, which affects its nucleation performance in PLA. Therefore, the modification of starch is to improve its nucleation performance in PLA, which is the key to whether it can be used as a high efficiency nucleating agent for PLA. Scholars mostly use thermoplastic starch, that is, to reduce dispersed phase size of starch in PLA through surface modification, and starch is an amorphous and highly plastic polymer in this form.
In addition, it can also provide a reaction group for starch to bind with PLA by modification, which more effectively improves the compatibility and nucleation effect of starch nucleating agent with PLA.
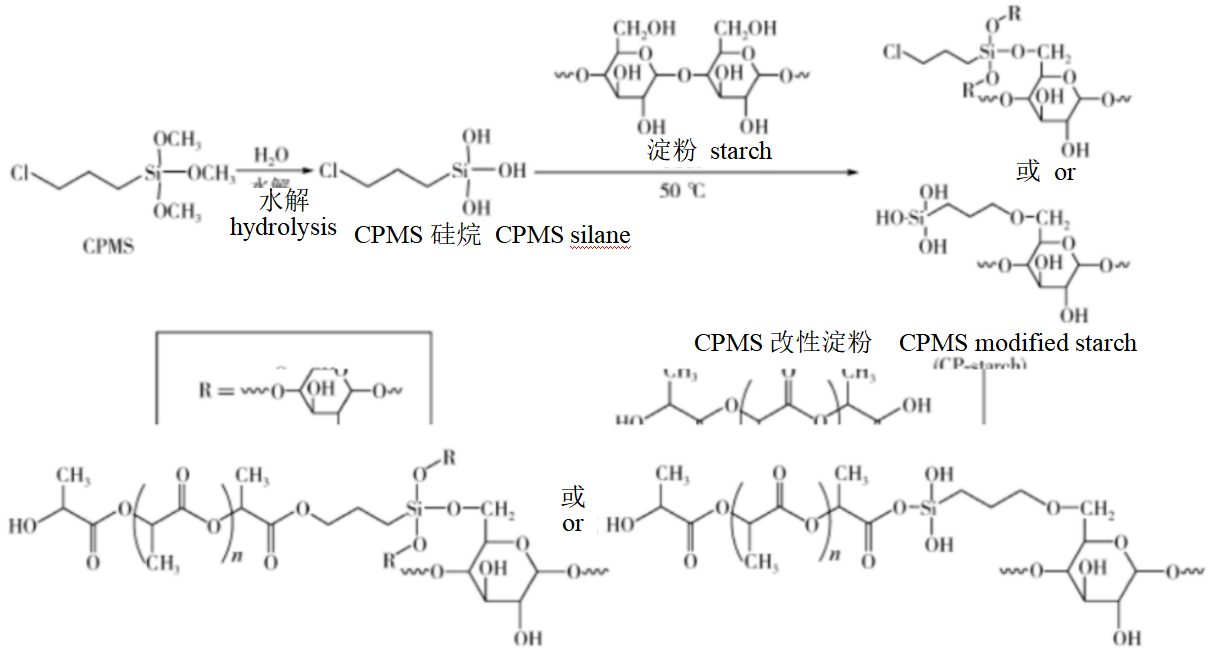
Figure 3: Schematic diagram of silane coupling agent modified starch and melt blend
Cellulose and cellulose nanocrystals: Cellulose is abundant in nature, and all plants and some tunicates, algae and bacteria can synthesize cellulose. Nanocellulose (NC) refers to cellulose with at least one-dimensional spatial size in the nanometer scale (1~100 nm), mainly including microfibrillated cellulose, nanocellulose crystals and bacterial nanocellulose.
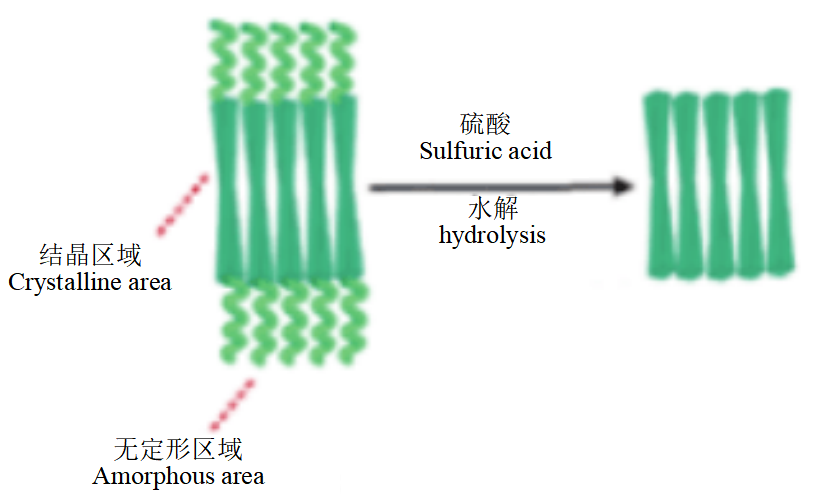
Figure 4: Schematic of preparation of cellulose nanocrystals
Cellulose can obtain rod-shaped Cellulose Nanocrystals (CNC) with high crystallinity by acidolysis and blending it with PLA can promote the nucleation of semi-crystalline PLA in the crystallization process and improve the crystallization speed of PLA, but the CNC itself is extremely easy to agglomerate. And the dispersion of CNC in PLA can be effectively improved by means of pre-solution treatment and surface modification.
Chitin: Chitin is the most abundant natural organic compound on Earth besides cellulose, and it is also the largest number of nitrogen-containing natural organic compounds on Earth other than protein, which widely present in the cell walls of crustaceans such as shrimp and crabs, insects, fungi, and plants. The chemical structure of chitin is similar to that of cellulose, with the basic structure being glucose units. The difference is that the hydroxyl group at the C2 position of cellulose is replaced by an acetamide group.
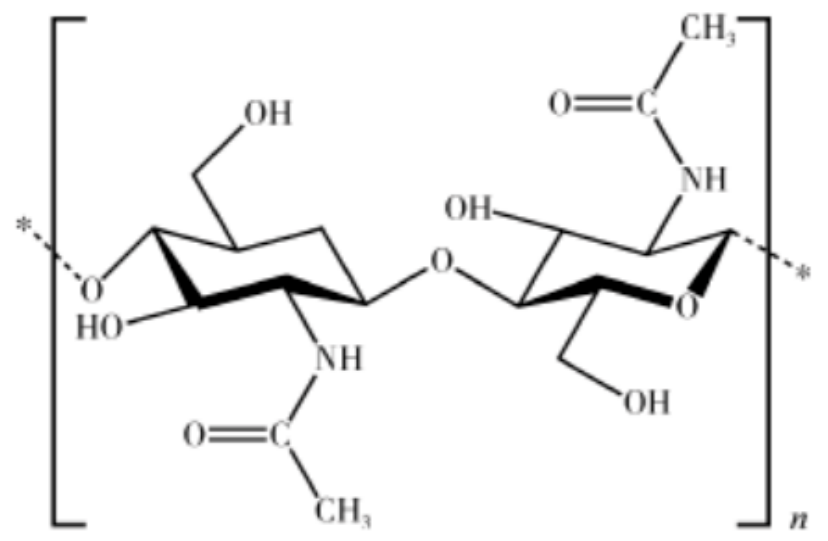
Figure 5: Structure formula of chitin
Chitin Whiskers (CHW) are individual chitin microcrystals obtained by acid lysis of chitin, which has excellent mechanical strength and modulus, and is an ideal polymer nanofiller.
Chitosan: Chitosan is the main component of the protective epidermis of crustaceans (such as crabs, shrimps, and lobsters) and the cell walls of some fungi (such as aspergillus and mucor) and can be obtained by de-ethylation of chitin.
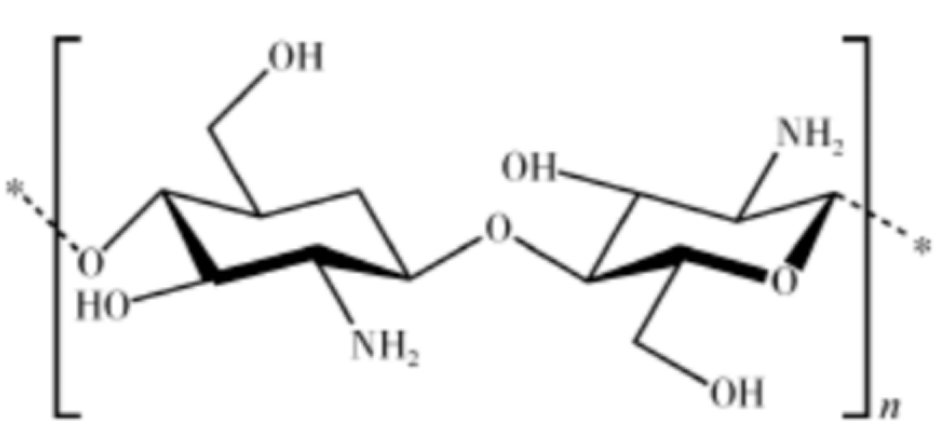
Figure 6: Structural formula of chitosan
Cyclodextrin: Cyclodextrin (CD) is a natural product of starch, a series of cyclic oligosaccharides produced by amylose under the action of cyclodextrin glucosyltransferase produced by bacillus, usually containing 6~12 D-glucopyranose units, of which more research and important practical significance is containing 6 , 7 and 8 glucose units of molecules, called α-CD, β-CD and γ- CD.
Unlike the above polysaccharide nucleating agents, CD is a class of supramolecular compounds with intraluminal hydrophobic, externally hydrophilic properties, capable of encapsulating polar compounds (such as alcohols, acids, amines and small inorganic anions) and non-polar compounds (such as aromatic and aliphatic hydrocarbons) in whole or in part.
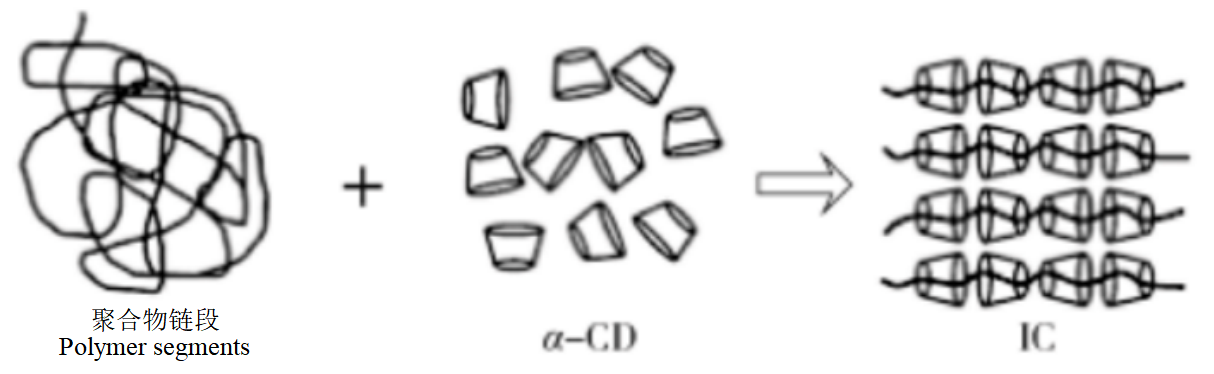
Figure 7: Schematic diagram of the process of clathrate formation
2. Phenolic Substances
Lignin: Lignin is a kind of cross-linked phenolic biopolymer in the cell walls of plants, which is a by-product of cellulose in the pulping process, producing over 70,000 kt of lignin annually. However, most of it is considered waste or used to generate heat. Lignin, due to its rich hydroxyl groups and intermolecular hydrogen bonding, easily absorbs moisture from air and aggregates, resulting in a dark brown wet powder state.
Figure 8 Schematic diagram of unimolecular chemical structure of lignin
3. Alcohols
Inositol: Inositol, also known as Cyclohexane-1,2,3,4,5,6-hexol, is currently commonly produced by pressurized hydrolysis using defatted rice bran as the main raw material. In theory, there are 9 possible isomers of inositol, all of which are isomers of glucose.
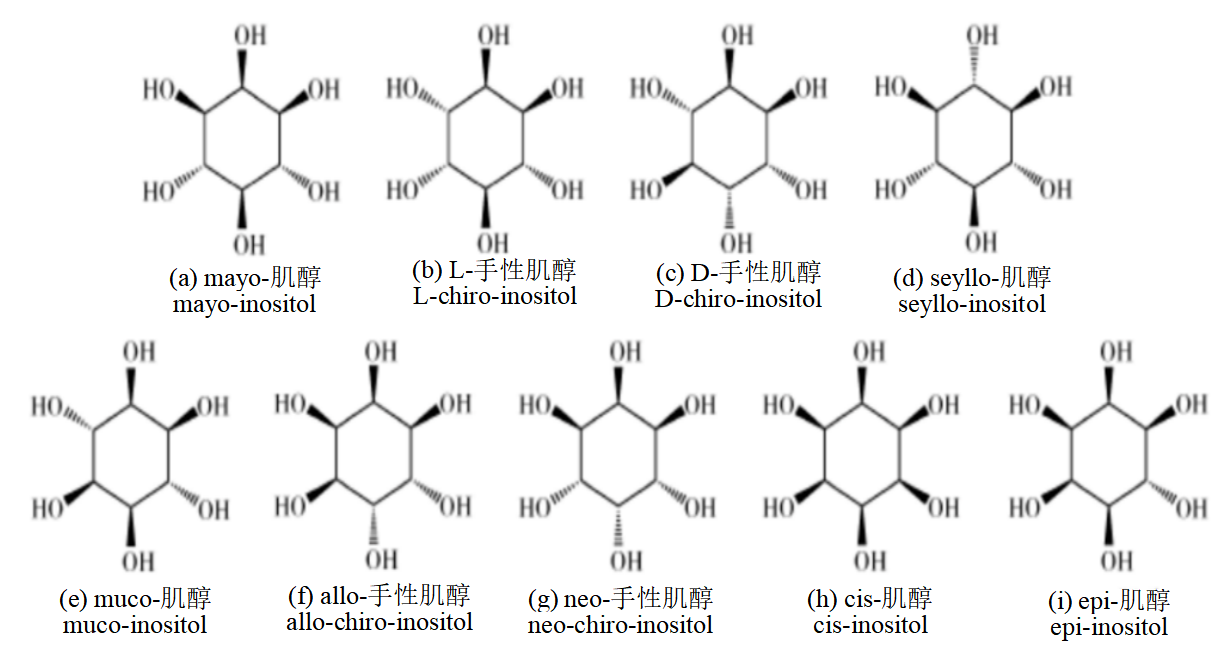
Figure 9: Molecular structural formula of 9 isomers of inositol
4. Proteins
Amino acids: Amino acids are the fundamental structural units of proteins in living organisms, with unique biocompatibility and complete bioabsorbability. Maria et al.’s research found that a small amount of polyglycine can significantly increase the crystallinity of PLA (60.5%), comparable to the effects of industrial talc (81.1%).
In addition, amino acid molecules become good ligands in chemical reactions due to their amino and carboxyl functional groups and can coordinate with a variety of metal ions to form amino acid metal salts.
5. Carboxylic Acids
Orotate Acid: Orotate acid (OA), commonly known as vitamin B13, is widely found in root plants and animal milk in nature, and is widely used in biomedicine, food and other fields.
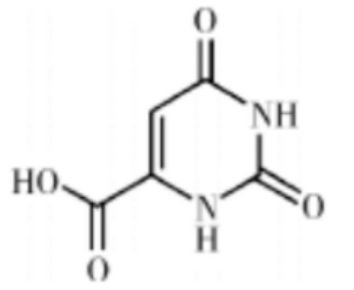
Figure 10: Molecular structural formula of OA
6. Nucleating Agents with Multibranched Polylactic Acid Structure
CN114634617A, CN114605625A discloses a class of organic nucleating agents for polylactic acid resin. The nucleating agent is a multibranched polylactic acid structure initiated and synthetized by hexamethylolmelamine. The end of the molecular chain forms an ester bond with one carboxyl group of the carboxylate group, while the other carboxyl groups and metal cations form ion bonds, and it is obtained by polymerize lactide, lactic acid, or polylactic acid; or obtained by polymerization of lactide initiated by hexamethylomelamine, when the reaction product reaching a certain molecular weight use oxalamide compounds for capping.
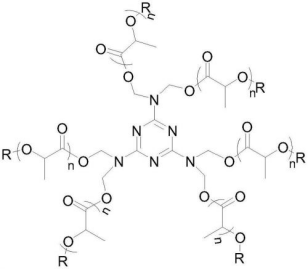
Figure 11: R represents carboxylate group and oxalamide group; the value of n is 50~500
Such organic nucleating agent has multiple branched polylactic acid molecular chains, and the dispersion in polylactic acid resin is better than that of general organic nucleating agents, whose grain size is close to the wavelength of visible light, capable of providing transparent resin composition; the branched chain structure of the organic nucleating agent is the same as that of the polylactic acid chain, which can make the molten polylactic acid molecular chain attached crystal growth under the appropriate temperature range, shorten the crystallization time and increase the heat deflection temperature of the polylactic acid resin. Nucleating agent and polylactic acid resin are synthesized from raw materials derived from plants in whole or most, and the plant degree of polylactic acid resin composition will not be reduced, and it is an environmentally friendly material.
The addition of such nucleating agents significantly increases the crystallization temperature and degree of polylactic acid resin. After fast annealing, the crystallization is complete, increasing the thermal deformation temperature of the product while maintaining its transparency.
Ⅴ. Conclusions
PLA has broad application prospects as a degradable and environmentally friendly material. Organic biomass nucleating agent has a wide range of sources and low costs, which can be matched with the biodegradable and recyclable properties of matrix PLA, and thereby meet the requirements of green environmental protection in the functionalized modification of PLA.
In the actual application, due to polysaccharides have been cheaper and easier to obtain, over the years, extensive and in-depth research has been conducted on thermoplastic starch, cellulose nanocrystals and other materials. The modification is mainly for the polyhydroxy structure caused by the nucleating agent and PLA poor compatibility problem, on the one hand by directly reducing the molecular size to improve the dispersion, on the other hand by introducing PLA chain segments and other surface functionalization modification means to improve the dispersion.
It is worth noting that future research work shall also start from the perspective of biodegradability in the selection of modification methods and modified substances, so as to conform to the green and environmental protection features of the system. The research on phenols, alcohols and carboxylic acid nucleating agents is relatively deficient, and although it can be proved to be an effective organic biomass nucleating agent for PLA, the specific crystallization mechanism needs to be further explored. Different from the aforementioned organic biomass nucleating agent, amino acids not only effectively promote PLA crystallization, but also catalyze lactide ring-opening polymerization in situ, which plays a significant role in the preparation and nucleation process of PLA, and worths further research.
Organic biomass has a wide range of sources, many categories, and a substantial number. Currently, the types of organic biomass nucleating agents used in PLA are relatively limited, and further research and exploration are needed on the purification, modification, and application processes of a large amount of organic biomass, which has an important impact on the wide application of PLA products.
Reference: CN114634617A、CN114605625A
“Research of Biomass Nucleating Agents for Polylactic Acid”, China Plastics
Previous:
Next:
Copyright: Yingkou Fengguang Advanced Material Co., Ltd.Liao ICP Filing No. 17015501-1 Technical Support:![]() onnuoIAD
onnuoIAD
© COPRIGHT 2021 FENGGUANG ADVANED MATERAL.ALL RIGHTS RESERVED